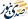
Promising Therapeutic Breakthrough for Chronic Inflammatory Bowel Disease
SadaNews - A multinational study has reached promising results for a new biological drug called "Guselkumab," which has demonstrated high efficacy in treating chronic inflammatory bowel disease, specifically moderate to severe active Crohn's disease.
Researchers, led by Mount Sinai Hospital in New York, USA, explained that the drug offers an effective and safe alternative, especially for patients who have not responded to traditional treatments; the results were published on Thursday in the journal "The Lancet."
Chronic inflammatory bowel disease, medically known as Crohn's disease, is one of the inflammatory bowel diseases that affect the digestive tract, primarily impacting the small intestine and colon.
The disease is characterized by chronic inflammation that leads to ulcerations and swelling in the intestinal walls, resulting in recurrent symptoms such as abdominal pain, chronic diarrhea, weight loss, and anemia.
It is one of the conditions that require lifelong medical management, with treatment efforts focusing on controlling inflammation, alleviating symptoms, and preventing complications such as bowel obstruction or the need for surgery.
Despite the availability of several biological treatments for the disease, many patients do not achieve sustained remission. The study relied on two phase three clinical trials that lasted 48 weeks.
The trials included more than a thousand patients worldwide, who were randomly divided into four groups to evaluate the efficacy of "Guselkumab" compared to another common treatment for Crohn's disease, "Ustekinumab," along with a placebo.
The results showed that "Guselkumab" achieved higher rates of intestinal healing and greater rates of attaining "deep remission of the disease," which is the state in which a patient is symptom-free with internal healing of the intestines.
The drug also demonstrated a significant ability to reduce dependence on corticosteroids, protecting patients from their long-term side effects, and it had a good safety profile, as side effects remained within expected limits without serious new issues. The drug "Guselkumab" has already received approval from the U.S. Food and Drug Administration based on these results.
Regarding its mechanism of action, the team notes that the "Interleukin-23" pathway is one of the key immune pathways playing a major role in promoting chronic inflammation associated with diseases like Crohn's disease. This pathway activates immune cells that secrete inflammatory substances contributing to tissue damage within the intestines.
"Guselkumab" specifically targets this pathway by disrupting the signals of the "Interleukin-23" pathway, helping reduce inflammation and heal the intestines without affecting other immune functions essential for the body.
The team confirmed that achieving the state of "deep remission of the disease" is associated with reduced flare-ups, the need for hospitalization, and complications such as surgery.
The researchers added that these results will influence how biological treatments are classified as first-line therapies for Crohn's disease globally, not just in the United States.

The Centenary of Idrees Al-Shraibi: A Biographer Who Explained the Defects of Colonialism...

A Spring for the Weary Heart.. When the Judas Tree Colors Istanbul

Magnetic Fields Around 7 Distant Worlds Open a New Window in the Search for Life

Ancelotti Leads the List: The 10 Highest-Paid Coaches in the 2026 World Cup

A Rare Recitation by Sheikh Mohamed Sidqi Al-Minshawi Returns to the Airwaves After Six De...

"I am your brother".. An Emotional Message from Younis Mahmoud to Iraq's Players Before th...

NVIDIA's RTX Spark Platform Ignites Laptop Prices